Chromatographic Method Validation
Course Details
In this webcast, our speakers present an introduction to chromatographic method validation. The session will consider topics such as why validate a chromatographic method, regulatory guidelines, eight steps of analytical method validation and a review of how and when to use statistical tests. A must see for everyone working in a regulated environment requiring validated methods or those wishing to benefit from the increased confidence that method validation brings.
Topics include:
- The method validation process
- Guidelines on implementing analytical method validation (AMV)
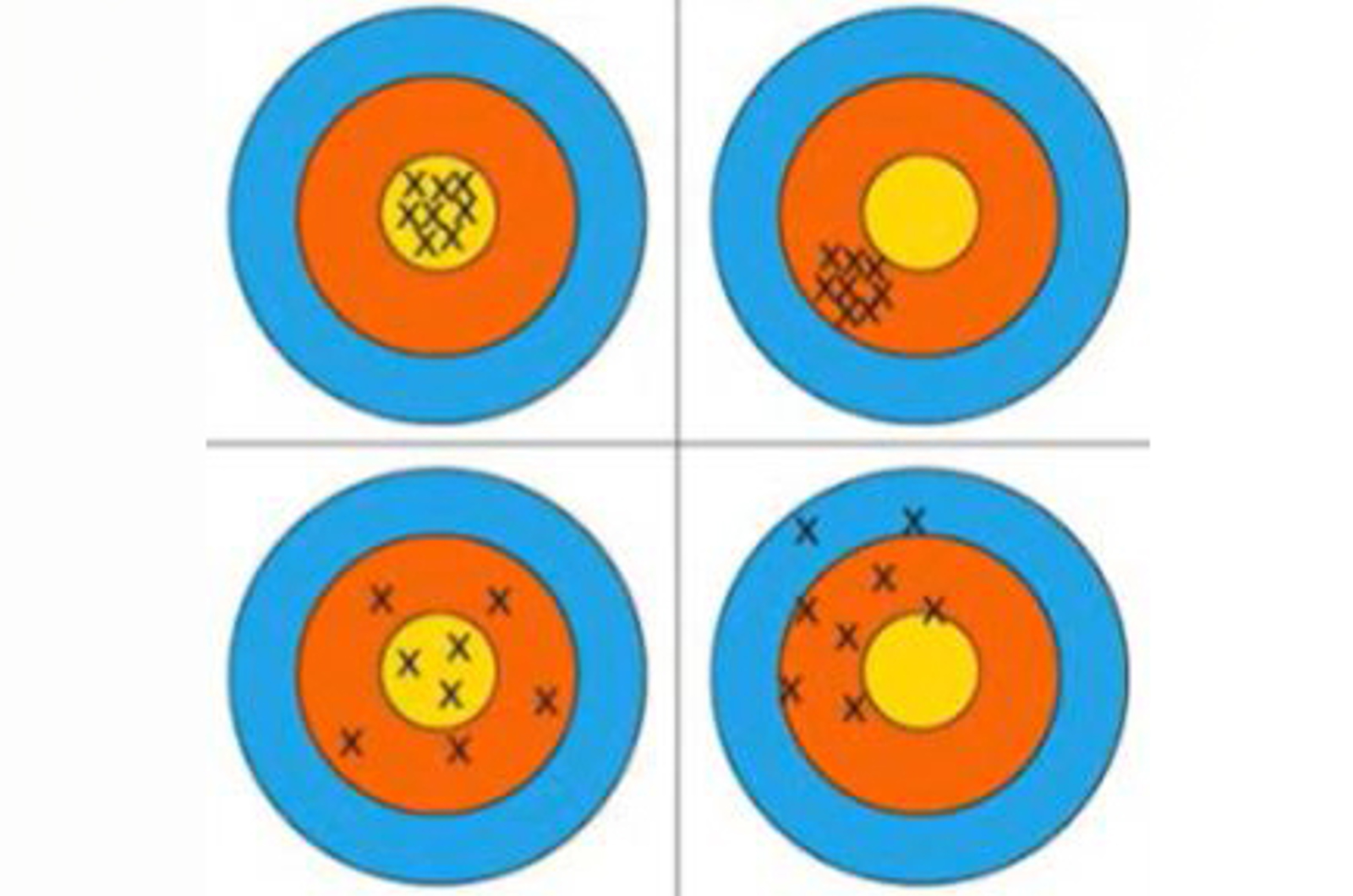
- Analytical performance characteristics - accuracy, limit of detection, limit of quantification, specificity/selectivity, linearity, range, robustness
- Statistical tests used in AMV